| People | Research |
|
Microflow Control by Exploiting Polymers as Functional Materials
Microflow control is critically important to biomolecular sensing and manipulation. We have been developing microflow control devices using polymers as functional materials. For instance, while polydimethylsiloxane (PDMS), an elastomeric polymer, is commonly used as microfluidic structural materials, we have exploited its large compliance to create innovative microfluidic control devices. As an elastomer, PDMS is highly compliant (with a nominal Young's modulus of 750 kPa, compared with 170 GPa for silicon). We have been purposely exploiting the compliance of PDMS to create microfluidic control devices. Structurally simple and relatively straightforward to fabricate, these devices represent innovative approaches to passive flow control, and offer interesting challenges in design and theoretical analysis. For example, we have developed microvalves and flow regulators that exploit the PDMS compliance. In the simplest configuration, these PDMS devices consist of a microchannel in which a flexible flap and a rigid block are arranged in series to form a flow constriction. 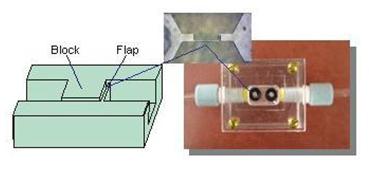
For valving applications, we have demonstrated a planar check valve that consists of a compliant thin flap that is perpendicular to the flow and in close proximity to or in contact with a rigid stopper. Fluid passage is allowed for forward flow by hydrodynamic force pushing the flap away from the restriction step, while reverse flow is blocked by pushing the flap against the stopper. Therefore, unidirectional flow can be achieved by the in-plane flap movement. The as-fabricated flap and stopper are in contact and form a zero-gap, i.e., a gap with no separation in the absence of flow. Testing results have shown that these planar check valves have negligible leakage rate, while forward flow rate increases rapidly with forward pressure. In particular, the valves shut off completely at reverse pressures higher than approximately 60 kPa, with leakage flow undetectable from the flowmeter used in the testing (detection limit: 1 l/min), corresponding to a diodicity (i.e., forward to reverse flow ratio) on the order of up to 105. 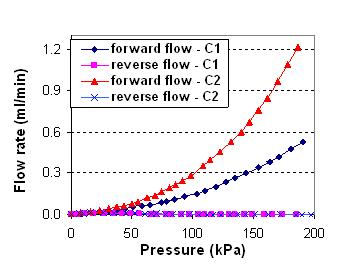
For flow regulation, the device is designed to function as a passively adaptive orifice. The fluid enters from the flap side of the channel, such that the flap is deflected toward the block, with which it effectively forms an orifice. The effect of an increase in the pressure across the device is to increase the flow rate and decrease the orifice size. The decrease in orifice size, on the other hand, has the opposing effect of reducing the flow rate. The flap-block system is designed such that these two effects cancel out to maintain a constant flow rate in the face of pressure variations. Testing results have shown that the micro flow regulator is capable of maintaining a constant flow rate with less than 1% fluctuations under pressures ranging from 100 to 200 kPa. 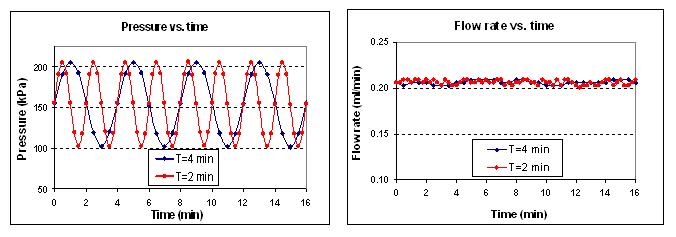
These compliance-based devices are currently being used as important enabling elements in the development of MEMS-based neurobiological drug delivery devices in a recently initiated collaborative effort with Professor Donald Pfaff at Rockefeller University. By allowing transdermal injection of drugs in small quantities over brief time pulses into brain tissue of laboratory mice, these devices will enable novel neurobiology studies that are not attainable with conventional tools.
|